|
Not all substances has a constant temperature of fusion, in fact a constant temperature is observed only for crystalline solids but not for amorphs (not crystalline) solids such as glass, wax, etc.Ģ. On the contrary, the heat which produces a temperature change is said sensible heat (line A-B).ġ. The heat which does not produce a temperature change is said latent heat of fusion (it is used to breakdown attractive forces between molecules and no temperature change is observed). Once all ice is transformed into water (point C) the temperature starts to increase again. By further addition of heat, until ice and water coexists, no change of temperature is observed (line B-C in figure). The temperature at which fusion begins (0 oC and 1 atm, in the case of water) is said fusion (melting) temperature. Let's consider the heating of ice (1 atm) from -10 oC to 0 oC, at beginning we can observe an increase of temperature and when the temperature reach 0 oC, fusion (melting) begins (formation of liquid water). Water’s melting point is 0☌, while its boiling point is 100☌.Fusion is the passage from the solid state to the liquid state and it requires heat (energy) to take place. Scientists refer to these as melting point, freezing point, condensing point and boiling point. These changes happen at precise temperatures for different substances. ice cubes shrinking in the freezer)Ħ) Deposition changes a gas to a solid. water forming on the outside of a cold glass)ĥ) Sublimation changes a solid to a gas. clothes drying on a clothesline)Ĥ) Condensation changes a gas to liquid. lake freezing over)ģ) Evaporation changes a liquid to a gas. dripping icicles)Ģ) Freezing changes a liquid to a solid. The six ways to change the phase (state) of matter:ġ) Melting changes a solid to a liquid. The movement of its molecules will increase until its physical state changes. When you heat a substance, you are adding energy to it. It is still the same matter or substance. 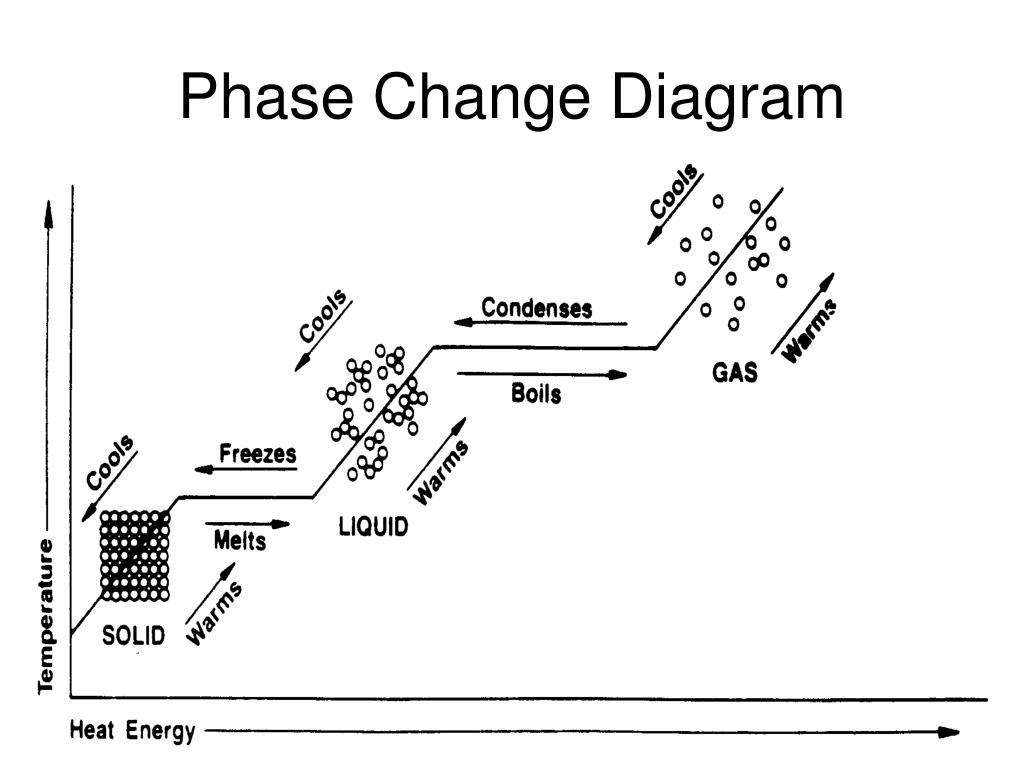
This does not change the matter’s molecular structure. increasing temperature or pressure) or releasing energy (i.e. Physical states can be changed by adding energy (i.e. Water is still H 20 when it is ice, steam or a liquid – even though its physical state has changed. Molecules can shift from one physical state to another without changing their molecular structure (or chemical state). These are physical states of the molecules of matter. There is a lot of free space between its particles and they flow easily past each other. Gas fills the shape and volume of the container in which it sits. There is not much space between the particles, but they can slide past each other and flow easily. Liquids have a fixed volume, but take the shape of the container in which they sit. There is not much space between the particles and there is little particle movement. A solid’s particles are packed closely together. 
Less commonly, we can also find matter as plasma or Bose-Einstein (BE) condensate. The three most common phases of matter on Earth are solids, liquids and gases. Matter can be found in several phases or states. Matter is something that has mass and volume (takes up space). 2) Name the three phases (states) of matter?ģ) Identify and describe the particle in each phase of matter and how they are different in each phase of matter.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
